Senior researcher, Professor Malka Cohen-Armon, describes the study she co-authored in Oncotarget entitled, “The phenanthrene derivative PJ34 exclusively eradicates human pancreatic cancer cells in xenografts.”

The Behind the Study series transcribes videos of researchers elaborating on their recent papers published in Oncotarget. Visit the Oncotarget YouTube channel for more insights from outstanding authors.
—
I am professor Malka Cohen-Armon, a senior researcher in Tel-Aviv University, Sackler School of Medicine. Our recent paper has been published in Oncotarget on November 22 (2019). Its title is The phenanthrene derivative PJ34 exclusively eradicates human pancreatic cancer cells in xenografts.
In this paper, we implemented a new mechanism we’ve recently identified of self eradication of human cancer cells. We developed human pancreatic tumors in immunocompromised mice and treated them for 14 days by a small molecule, PJ34. A month later, without any additional treatment, we extracted the tumors and measured a drop of about 90% of the pancreatic cancer cells in the tumors of the treated mice relative to the cancer cells in tumors of untreated mice. In one mouse, the tumor disappeared completely. The treatment did not adversely affect the development of the mice. They continued to gain weight as untreated or control mice.
As I mentioned, the current research was based on a mechanism we discovered two years ago. It was also published in Oncotarget. In general, I’m interested in unveiling unknown signal transduction mechanisms in diverse research field. I was once focused on the understanding of signal transduction mechanism implicated in long-term memory acquisition. In that research, we identified anti-epigenetic mechanism mediated by the activation of a nuclear binding protein part one, which is mainly known for its role in DNA repair. This finding drew our interest to the role of this epigenetic mechanism in immediate early gene expression, implicated in a variety of similar processes under physiological and pathological conditions.
The findings of that research were published in science, in the Journal of Cells in Nature. An incidental finding during this research unveiled the unusual activity of a group of small molecules with the chemical structure. These small molecules were designed as PARP inhibitors to protect quiescent cells like neurons and cardiomyocytes from cell death under stress conditions like stroke.
It was unexpected to find that these same molecules are highly toxic in human cancer cells while not affecting normal proliferating cells. This finding attracted our attention and later on the attention of other researchers because of its potential role in cancer therapy. So we started digging into the phenomena in an attempt to find the underlying molecular mechanism discriminating cancer cells from normal proliferating cells. The information we gathered led to a mechanism associated with mitosis in the treated human cancer cells. It turns out that human cancer cells treated with these molecules were eradicated during mitosis due to an anomaly is a construction of their mitotic spindles. This anomaly was not induced by the same treatment in normal sense. Furthermore, we identified specific proteins implicated in mitosis that are targeted by the most potent molecule in this group, PJ34. This molecule prevented the clustering of NuMA in the spindle pole.
NuMA is an acronym for Nuclear Mitotic Apparatus Protein, and it is a protein necessary for mitosis. Actually, mitosis is prevented by NuMA silencing in any cell; we found that PJ prevents the post-translational modification of NuMA only in human cancer cells, modifications by phosphorylation of NuMA at a specific site, and PARsylation of NuMA by tankyrase 1.
Preventing NuMA’s post-translational modification prevents its ability to bind to other proteins and this prevents the translocation of NuMA to the spindle pole and its clustering in the poles. NuMA is a substrate of serotonin kinase P1 and tankyrase 1. Both proteins are inhibited by PJ34 and are hardly expressed in normal cells. So the post-translational modification of NuMA is required for the binding of NuMA to proteins.
NuMA binding to proteins is required for its translocation and clustering in the spindle pole and the clustering of NuMA in the spindle poles constructs and stabilizes the poles, a feature required for the segregation and the alignment of chromosomes in the spindle mid-zone. Chromosomes alignment in the spindle mid-zone guarantees a normal separation of the doubled chromosomes in the dividing cell equally divided between the two daughter cells. An anomaly, preventing mitosis is not tolerated in any cell.
The dispersion of the chromosomes in the dividing cell causes mitosis (inaudible) in this step of division the cells end by eradication. This fast eradication is by a cell death mechanism called mitotic catastrophe cell death.
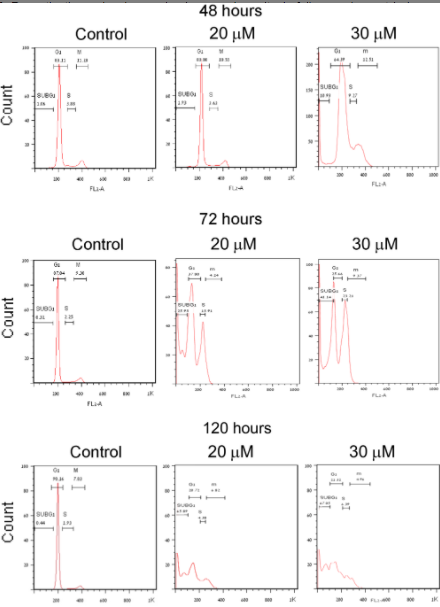
This suggested the molecular mechanism for the toxicity of PJ34 in human cancer cells was stressed by confocal microscopy. In cancer cells treated with PJ34, immunostained NuMA and stained chromosomes were both dispersed in the mitotic spindle. This did not occur to normal clustering and chromosome segregation in normal proliferating cells treated with PJ34. The process of cell death during mitosis lasts minutes, and therefore it can be filmed online by confocal microscopy. It has been documented in triple negative breast cancer cells in our paper in JoVE in 2013. This cell death mechanism differs from apoptosis which may last for hours. So in summary, PJ causes an anomaly which prevent the segregation of the chromosomes and their alignment in the meet zone of the mitotic spindle in human cancer cells, the dispersion of chromosomes in the mitotic spindle, arrest mitosis in the anaphase.
This anomaly is not tolerated by control mechanisms existing in every cell and instead of division, the cells are self-eradicated. We identified this mechanism in cancer cells that are resistant to cancer therapy, including triple negative breast cancer cells, pancreatic cancer cells, lung cancer cells, and glioblastoma. The more frequently cells started to divide, the more frequently they die. This is probably the reason for the eradication of variety of human cancer cells in by PJ34, within 72 to 96 hours. In the current paper, the toxic activity of PJ34 was tested in the human pancreatic mouse develop immunocompromised mice. It has not been done in humans. It is a pre-clinical study. It was during this study, it was challenging to find the target proteins of PJ34 among the many proteins known to be involved in mitosis.
It was rewarding on the other side to see that PJ34 acting by the mechanism we disclosed actually kills human cancer cell without impairing normal cells, which all cause trauma and they are very frequent and apparently 50% of the tumors in pancreatic cancer tumor. Furthermore, it was rewarding to find that the treatment with PJ34 does not interfere with the way to gain development and behavior of the treated mice.
We next intend to test the potential of the molecule PJ34 for cancer therapy. We measure its pharmacokinetics and bioavailability and for now PJ doesn’t seem to be toxic to mice, however, safety has to be fully tested. We have to follow the FDA regulations and perform all the experiments that are required for drug development. This might last at least two to three years for before being able to start clinical trials. We hope that this novel therapeutic mechanism will provide a new mode of cancer therapy for cancers resistant to currently used therapies.
Click here to read the full study published in Oncotarget.
—
Oncotarget is a unique platform designed to house scientific studies in a journal format that is available for anyone to read—without a paywall making access more difficult. This means information that has the potential to benefit our societies from the inside out can be shared with friends, neighbors, colleagues and other researchers, far and wide.
For media inquiries, please contact media@impactjournals.com.



