Researchers use a computer simulated modeling system to highlight the strengths and weaknesses of two ALK inhibitors.
The Trending with Impact series highlights Oncotarget publications attracting higher visibility among readers around the world online, in the news, and on social media—beyond normal readership levels. Look for future science news and articles about the latest trending publications here, and at Oncotarget.com.
—
Despite many therapeutic advances over the years, over half of patients with lung cancer die within one year of diagnosis. Non-small cell lung cancer (NSCLC) comprises 85% of all lung cancer, and around 3–7% of patients with NSCLC present with a rearranged ALK gene (ALK+). This abnormality produces aberrant ALK protein cell signaling pathway activity and causes cancer cells to grow and metastasize. ALK+ NSCLC patients often develop drug resistance to available ALK inhibitor drugs.
“Consequently, it is of the upmost importance to adequately use the currently available treatments in the correct order to maximize the life span of NSCLC patients.”
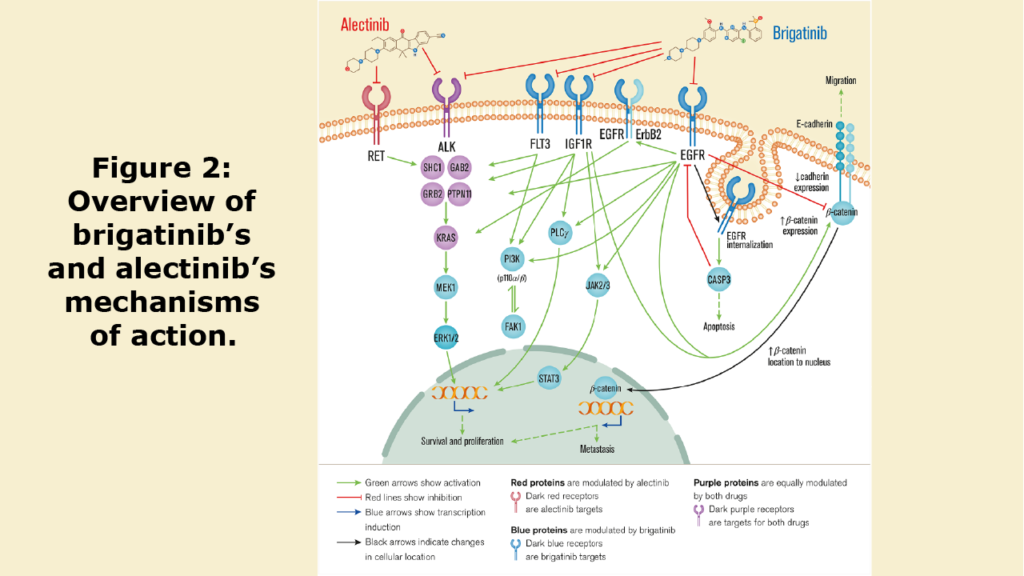
In 2021, researchers from Spain conducted a study published in Oncotarget, titled: “Head to head evaluation of second generation ALK inhibitors brigatinib and alectinib as first-line treatment for ALK+ NSCLC using an in silico systems biology-based approach.” This trending paper was authored by researchers practicing at the Hospital Germans Trias i Pujol, Takeda Farmacéutica España, Anaxomics Biotech, and Universitat Pompeu Fabra.
The Study
The researchers first began their study by characterizing the pathophysiology of ALK+ NSCLC after completing a detailed review of review papers published in PubMed between 2013 and 2018.
“To carefully characterize the pathophysiology of ALK+ NSCLC, we conducted an extensive and detailed full-length review of relevant review articles over the last 5 years in the PubMed database (from December 3rd 2013 to December 3rd 2018)[…]”
Next, to compare the strengths and weaknesses of two second generation ALK inhibitor drugs, brigatinib and alectinib, the researchers used a computer simulated modeling system—in silico. They explain that an in silico method of study can be highly useful when analyzing drug characteristics and predicting the biochemical characteristics and drug mechanisms of action.
“Overall, these systems can be employed for the exploration of anticancer drug mechanisms of action and their efficacy in specific patient profiles.”
The in silico system they used is called a Therapeutic Performance Mapping System (TPMS) and is based on artificial intelligence and pattern recognition models. This TPMS system was “trained” by the researchers in this study and given up-to-date biological and clinical data to input into its configuration. The mathematical models used to obtain the ALK inhibitors’ mechanisms of action were generated following the same methodology as described in this study.
“This methodology integrates available biological, pharmacological and medical information to generate mathematical models that simulate the mechanisms of action of drugs in a pathophysiological human context (Figure 4).”
To detect and explain the biological relationships that occur, the team used two distinct modeling methods: artificial neural networks and sampling-based methods. They applied Sobol sensibility analysis over the TPMS mathematical models in order to account for the impact of any noise affecting the final mechanisms of action. The researchers also performed drug-(patho)physiology motive relation finding and evaluated the impact of potential resistances and drug interferences over the mechanisms of action.
Results & Conclusion
“According to the current knowledge and the data herein presented, brigatinib might be more prone to present relevant metabolic and mechanistic interactions with other drugs than alectinib, which might be a safer option in poly-treated patients.”
“Brigatinib appears to have a wider mechanism of action, presenting targets that potentially act more strongly in most of the ALK+ NSCLC pathophysiological pathways, including invasiveness to the CNS [central nervous system].”
“On the other side, alectinib-induced RET inhibition might contribute to reducing the tumour immune evasion mechanisms.”
The researchers found that both drugs are known to be well-tolerated and show similar efficacy for the treatment of ALK+ NSCLC in a first-line setting. However, they explain that the differences in their characteristics shown in this study might allow for administration in more targeted patient populations that might see benefits from either brigatinib or alectinib. This deeper classification may also help when considering potential safety concerns in specific patient subpopulations.
“Future clinical studies will be needed to confirm these findings. The used approach can be applied for the evaluation of other next-generation ALKi, even if not yet approved, or exploring other questions, such as optimal treatment sequence.”
Click here to read the full scientific study, published in Oncotarget.
—
Oncotarget is a unique platform designed to house scientific studies in a journal format that is available for anyone to read—without a paywall making access more difficult. This means information that has the potential to benefit our societies from the inside out can be shared with friends, neighbors, colleagues and other researchers, far and wide.
For media inquiries, please contact media@impactjournals.com.



