Researcher Mary Janatpour describes the findings in a paper she helped authored in Oncotarget, entitled “Low-dose metronomic cyclophosphamide complements the actions of an intratumoral C-class CpG TLR9 agonist to potentiate innate immunity and drive potent T cell-mediated anti-tumor responses.”

The Behind the Study series transcribes videos of chosen researchers elaborating on their recent papers published in Oncotarget. Visit the Oncotarget YouTube channel for more insights from outstanding authors.
—
Hi, my name is Mary Janatpour, and I’m currently Senior Vice President of Biology at Spotlight Therapeutics in the Bay Area. The work that we’re going to discuss today is work that was conducted in my laboratory while I was vice president of oncology research at Dynavax Technologies. The paper that we’re discussing is entitled Low-dose metronomic cyclophosphamide complements the actions of an intratumoral C-class CpG TLR9 agonist to potentiate innate immunity and drive potent T cell-mediated anti-tumor responses.
So, as we appreciate in the field of cancer therapy, immune therapies have given us a leap forward with checkpoint blockade conferring durable responses in a subset of patients. And so this is a big breakthrough in the field, but it is appreciated by the field that in order to reach those numerous patients that are not impacted by the checkpoint blockade monotherapies, that we need to identify rational combinations to increase efficacy, ideally without introducing additional toxicity.
So, at Dynavax Technologies, our focus was on TLR9 agonism, and specifically what we did here was develop a class C-class of oligonucleotides that optimally activates TLR9 in plasmacytoid dendritic cells. The importance of this is that while checkpoint blockade takes the breaks off T cells, we need T cells to act on. And by stimulating plasmacytoid dendritic cells, we can activate the innate immune system to prime T cells on which checkpoint blockade can act. Furthermore, with the TLR9 agonist, what we learned is that to optimally activate them to confer this efficacy, we need to administer them intratumorally. So, the TLR9 agonist are administered intratumorally. This then in juxtaposition with tumor antigen, and now we can optimally prime a T-cell response to then combine with a checkpoint blockade.
Work by my colleague, Cristiana Guiducci, was foundational in showing that intratumoral administration of TLR9 agonist, specifically these class C agonists, combined with checkpoint blockade, conferred efficacy and cured mice of tumors. And we have since demonstrated in the clinic that this combination is quite efficacious. However, there are still patients that will not respond to this doublet.
So, what we undertook in my laboratory was to try to identify clinical compounds that could be rationally combined with this doublet to confer increased efficacy, again, without additional toxicity. And so we looked to clinical agents, and there were a number of classes of compounds that could be rationally combined with TLR9 agonism. For the context of this paper, we focused on chemotherapies. Now chemotherapies at high doses actually are quite toxic, although they do confer some immune modulatory effects such as generating immunogenic cell death. But our interest was to look at them at low doses, because there is a fairly significant literature showing that low-dose chemotherapies can have immunomodulatory effects that would be predicted to be complimentary to our TLR9 agonist.
We did a thought exercise and then a small screen to hone in on where we wanted to focus, and we landed on a low-dose metronomic cyclophosphamide as an appropriate combinatorial candidate to complement the activities of our class C TLR9 agonists to optimally generate an immune response.
Now, it should be noted that a low-dose cyclophosphamide has been in the clinic for many years, originally for anti-angiogenic properties and more recently appreciated for its ability to decrease regulatory T cells. Also, others have combined low-dose cyclophosphamide with TLR9 agonists. All those other TLR9 agonists have been, what we call, class B TLR9 agonist. So, in our class C TLR9 agonist, these are optimal activators of TLR9, in that they both induced interferon and mature the plasmacytoid dendritic cell, in contrast to other classes which kind of do one or the other.
So, what we did in this paper, which I think adds to our understanding, was not only did we demonstrate that the combination very potently regresses tumors, but we added some mechanistic insight into what’s happening IN what we call the tumor microenvironment. So, tumors orchestrate their microenvironment to be immunosuppressive, such that even when we have a T-cell priming response like we get with giving our TLR9 agonists with a take-off-the-brakes on the T cell with checkpoint blockade, there’s sill suppression in that environment. So, what we looked at was the ability of this combination to modulate the tumor micro environment, to make it more immune permissive.
So, while in low-dose cyclophosphamide it has been demonstrated to decrease Tregs and do some good things, one of the knocks against it, and in the hypothesis, is that it also has been shown by some people to increase, what we call, monocytic myeloid-derived suppressor cells. And these would be predicted to be immune-suppressive. When we looked at the tumor micro environment and what was going on, we were very delighted to see that, in fact, what we’re observing in our models is that the low-dose cyclophosphamide is actually decreasing mature TAMs, mature tumor-associated macrophages, and increasing a population that are high in MHC class II, and appear to be increased in their overall increasing of the antigen processing in the tumor.
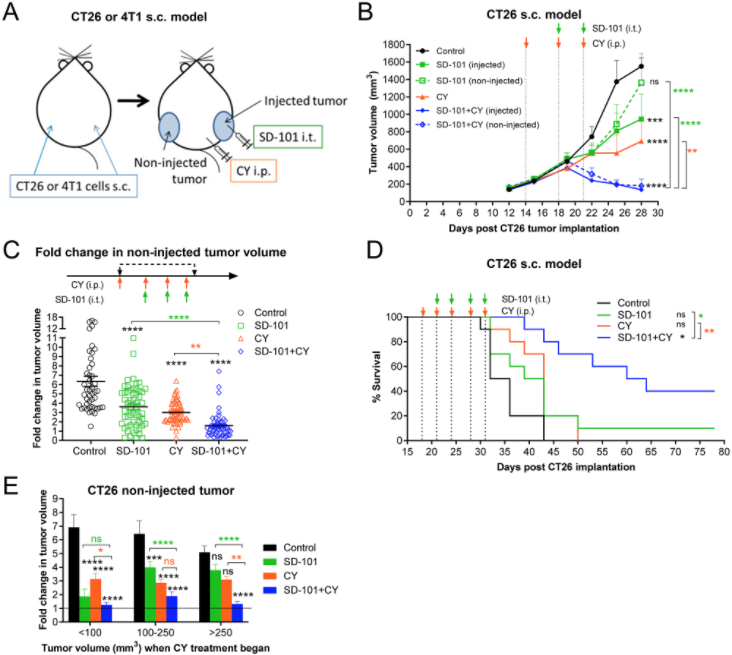
So on balance, if in fact low-dose cyclophosphamide is inducing any suppressive effects, on balance, the anti-tumor effects, the immune permissive effects, are actually dominating the picture. So, that was very encouraging.
The other insight, I think, our paper brings to the field, is that the kinetics of the changes in the Tami are quite rapid. So, when after only the first dose of the combination of both SD-101, our TLR9 agonist, and low-dose cyclophosphamide, after the very first dose, we actually see dramatic changes in the tumor microenvironment to confer this anti-tumor response.
Additionally, what was encouraging to see, was that when we stopped the combination treatment, those changes in the tumor microenvironment and the immune responses actually are persistent. So, I think the kinetics and understanding of what’s going on with the tumor microenvironment in a global setting is what this paper adds to our understanding of combining low-dose cyclophosphamide with TLR9 agonists.
What we would want to do next to better understand what’s going on, is to really dissect the functional components of each of these immune subsets to see what their contribution is. In the paper, we demonstrate that, ultimately, the anti-tumor responses are being mediated by CD8+ T cells, that there is a memory response, and all those things that we want to see, but to get there, I think there’s more work to do to understand how the modulation of the different immune compartments is getting us to that point. So, biology standpoint, that’s, I think, what needs to be done next to follow on our observations presented in this paper.
And then at bigger picture, the reason why we executed these studies was to inform clinical studies to improve clinical efficacy. And so, I think our paper, together with other similar papers, suggests that adding low-dose metronomic cyclophosphamide, which is quite feasible because it’s a generic drug that is given orally as a pill, adding low-dose metronomic cyclophosphamide to a regimen of localized, innate stimulation plus checkpoint blockade, could confer significant benefit to patients. And so my hope is that this data will be considered in that light and may inform clinical studies that are actually happening now and could possibly benefit from adding this regimen.
I invite you to read some of the foundational research that was done by my colleague, Cristiana Guiducci, and her group in setting this up, and to be on the lookout for clinical papers of are localized TLR9 agonist, plus checkpoint blockade, which are some are published and some are coming out. And then for the clinicians out there, to consider adding low-dose metronomic cyclophosphamide to that regimen of localized innate stimulation, together with the checkpoint blockade. There are some studies right now with low-dose cyclophosphamide and checkpoint blockade, but I think it’s really important that the localized innate stimulation be a part of that package. So, thanks.
Our experience working with Oncotarget has been quite positive. We chose this journal because of its scope, and we thought that our work was a very solid piece of science and that it would fit very well into the scope of Oncotarget. We were very pleased with the rigorous review of our document.
Our manuscript, we got good feedback from our reviewers that we were able to incorporate into the manuscript to improve it. The logistics and communication were quite optimal, in the sense that there we were dealing with real people in our correspondence with some of the back-and-forth.
So, I view Oncotarget as a very valuable journal. I think there are a lot of key publications that I have turned to in it as a journal and I feel very privileged that we are able to publish in Oncotarget, and it’s been a very positive experience.
—
Oncotarget is a unique platform designed to house scientific studies in a journal format that is available for anyone to read—without a paywall making access more difficult. This means information that has the potential to benefit our societies from the inside out can be shared with friends, neighbors, colleagues and other researchers, far and wide.
For media inquiries, please contact media@impactjournals.com.



