A team of US researchers sought to halt tumor growth in breast cancer by targeting breast cancer cell metabolism in the mitochondria through the use of an adenosine triphosphate (ATP) synthesis inhibitor.
The Trending with Impact series highlights publications attracting higher visibility among readers around the world online, in the news, and on social media — beyond normal readership levels. Look for future science news and articles about the latest trending publications here and at Oncotarget.com.
—
Breast cancer has been top-of-mind for many researchers over the years. Some focus on early detection methods and others set their sights on discovering new treatment options — both are crucial for patient survival.
The innumerable potential mechanisms researchers may target using countless variations of therapeutic treatments for breast cancer is vast, and, the nature of human biology sees to it that for every action, there are reactions. Some of these reactions require multiple studies that reveal links between genes in our DNA that were previously undiscovered.
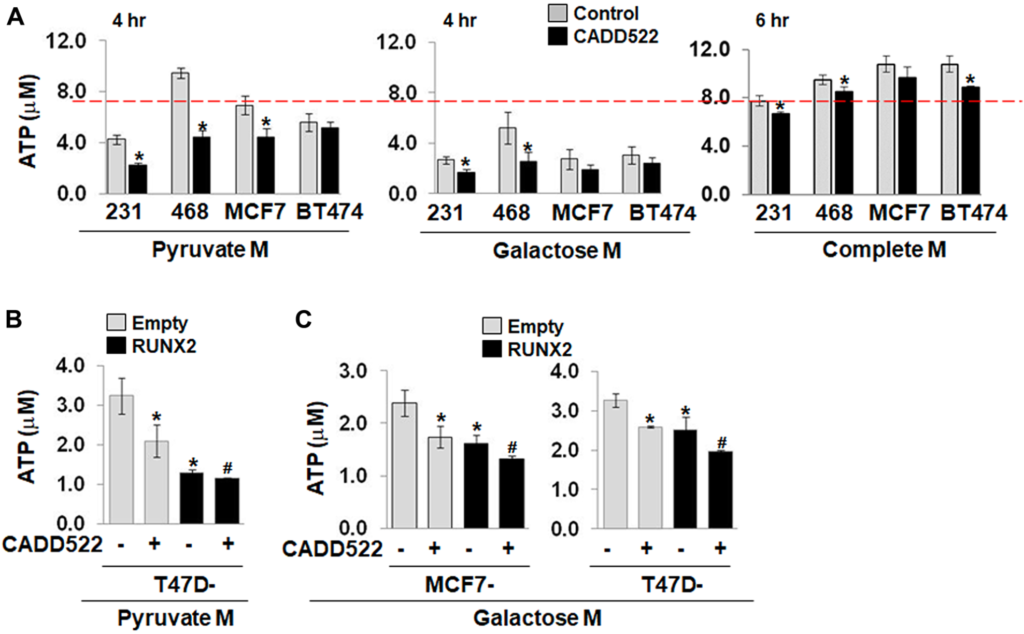
One such study was published recently in Oncotarget and conducted by a multidisciplinary team of skilled researchers from the United States’ Maryland and California. The team sought to halt tumor growth by targeting breast cancer cell metabolism in the mitochondria through the use of a new adenosine triphosphate (ATP) synthesis inhibitor.
ATP is a critical molecule in many human cell processes, including energy storage and transfer, cell signaling, cellular respiration, and cell metabolism. CADD522 is a small and potent molecule used as a drug to inhibit the RUNX2 transcription factor, which is well-known to promote breast cancer growth and metastasis.
“Cancer stem cells are slow-growing cells that harbor tumorigenic potential, self-renewal capabilities, and intrinsic resistance to conventional and targeted therapies. These cells are dependent on active mitochondria and show increased oxygen consumption and ATP production compared to more highly proliferating tumor cells [100, 102–104].”
Given cancer stem cells are dependent on active mitochondria, the team proposed that the inhibition of the mitochondrial respiratory chain can be used to develop selective anticancer agents. The study targeted mitochondrial metabolism by inhibiting cell respiration in breast cancer cells and reported that CADD522 promotes growth arrest, not apoptosis.
“CADD522 diminished ATP levels through inhibition of mitochondrial ATP synthase activity, without alteration of complex I activity.”
The researchers studied the reactions of control and breast cancer cell-lines in combination with CADD522 and other agents through a complex set of assays, scans, statistical analyses, and other tests.
Conclusion
Through their findings, the team notes that they do not exclude the possibility of CADD522 indirectly interfering with mitochondrial dynamics through regulation of nuclear genes that target the processes that regulate mitochondrial content, structure, and function.
“Further studies will be needed to verify the precise effects of CADD522 on regulation of mitochondrial biogenesis.”
The study results demonstrate that mitochondrial ATP synthase inhibition may be a valid therapeutic approach and that the suppression of mitochondrial metabolism is indeed a mechanism that drives sensitivity of breast cancer cells to CADD522.
Click here to read the full scientific paper, published in Oncotarget.
—
Oncotarget is a unique platform designed to house scientific studies in a journal format that is available for anyone to read – without a paywall making access more difficult. This means information that has the potential to benefit our societies from the inside out can be shared with friends, neighbors, colleagues and other researchers, far and wide.



