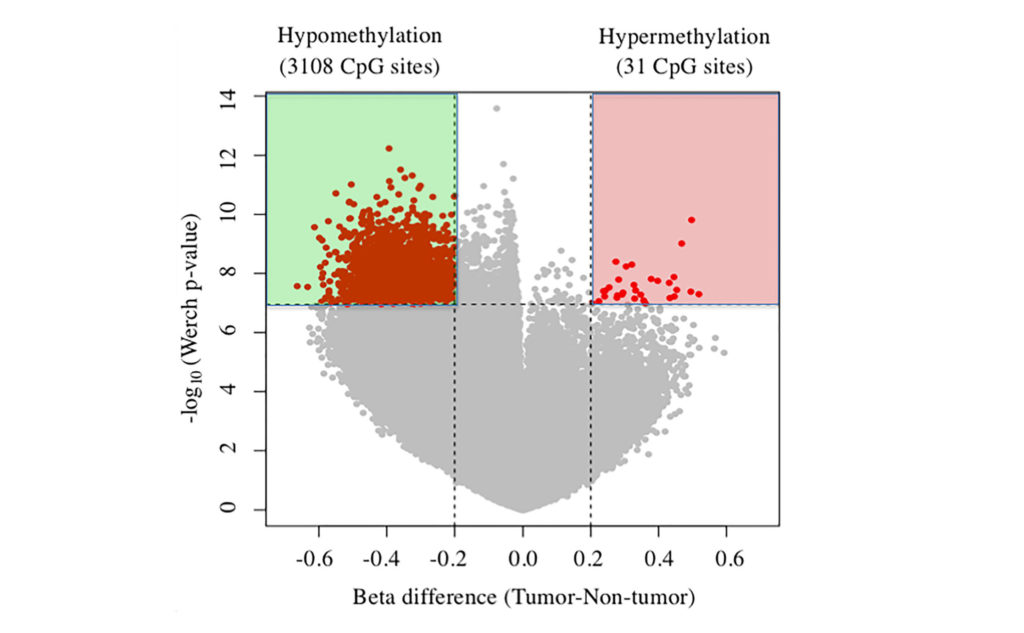
A genome-wide analysis of DNA methylation concluded that the TBX15 gene is hypermethylated and downregulated in tumors of patients with hepatocellular carcinoma.
Hepatocellular carcinoma is considered rare, though, it is also the most common primary tumor of the liver. Fortunately, most patients do not suffer from symptoms in the earlier stages of this disease. Unfortunately, patients diagnosed with late-stage hepatocellular carcinoma often present with symptoms caused by an underlying liver disease.
“Hepatocellular carcinoma (HCC) is the sixth most prevalent human cancer and third-highest cause of cancer-related deaths worldwide, and its incidence is significantly increasing [1, 2].”
Due to the high rate of post-treatment HCC relapse and metastasis, researchers based out of Japan—from the Institute of Biomedical Sciences at Tokushima University, Kumamoto University, and Kyushu University Beppu Hospital—conducted a full-genome analysis of DNA methylation in tumor and non-tumor HCC tissue samples in search of a new prognostic biomarker with potential use in clinical treatment.
DNA Methylation
Methyl groups are small, stable, non-reactive molecules that consist of one carbon and three hydrogen atoms. They are naturally introduced in the DNA at various times—for a number of reasons. In short, the binding and enzyme-mediated functions of methyl groups are intended to ensure biological processes and genes within the DNA operate correctly. Although they are non-reactive, methyl groups can alter gene expression through epigenetics, or changes caused by the modification of gene expression (not in genetic code). Methylation reactions are crucial to processes such as gene expression, neurotransmitter synthesis, and liver detoxification. Researchers use hypermethylation in DNA as a potential indication of erroneous gene functioning, which may contribute to cancer propagation.
The Study
Previous studies note that there is a biased expression of the TBX15, or T-Box 15, gene in the liver. Studies have also found that TBX15 is downregulated in breast, prostate, ovarian and other cancers, suggesting additional evidence that T-Box 15 may be used as a prognostic biomarker in some cancers.
“Moreover, evidence indicates the association of diverse T-box genes in oncogenesis, tumor invasion, and metastasis of certain cancers [19, 20].”
The team in this study conducted a genome-wide analysis of DNA methylation in tumor and non-tumor tissue from 15 patients with hepatocellular carcinoma, analysis of messenger RNA (mRNA) levels in T-Box 15 genes of a 371 hepatocellular patient dataset obtained from the Broad Institute’s Firehose, and analyzed a validation set composed of 58 patients with HCC who had radical liver resection between April 2004 and March 2011 at the Tokushima University Hospital in Japan. They then analyzed and investigated the relationships between T-Box 15 mRNA expression and the patient’s tumor phenotype.
“Tumor status, stage, and curability were defined according to the Classification of Primary Liver Cancer by the Liver Cancer Study Group of Japan.”
A variety of multi- and univariate analyses were conducted, including microarray analysis of DNA methylation, immunohistochemistry staining, quantitative real-time reverse transcription-PCR to evaluate mRNA levels of TBX15 in tumor and non-tumor liver tissues, TCGA methylation analysis of HCC, TCGA pan cancer analysis of mRNA expression, gene set enrichment analysis, and statistical analysis.
Results
In their analysis of mRNA expression in T-Box 15 genes from HCC tumor tissue, the researchers found that levels were significantly lower than in non-tumor tissue from HCC patients.
“In our validation set comprising 58 patients with HCC, we found that TBX15 mRNA expression was significantly reduced in tumor tissues compared with that of non-tumor tissues, likely because of TBX15 hypermethylation.”
They found that TBX15 expression did not significantly correlate with the status of the tumor, except in levels of serum DCP. Another significant finding was that the relatively low expression of TBX15 in tumor tissue was an independent prognostic factor for overall and disease-free survival. The team also observed a negative correlation between the reactive oxygen species (ROS) pathway and TBX15 expression.
“Particularly in the tumor microenvironment, moderate ROS concentration induces cancer cell survival, angiogenesis, and metastasis via several cell survival signaling cascades, such as MMPs and VEGF, meanwhile, high ROS concentration leads to cancer cell apoptosis [39].”
Conclusion
The team revealed that TBX15 was the most hypermethylated gene in tumor tissue samples compared with non-tumor tissue samples, and that TBX15 mRNA levels in tumor tissues were significantly lower compared to non-tumor tissues.
“In HCC with low TBX15 expression, ROS production could be moderate and contribute to a worse tumor malignancy.”
Given their findings, the researchers in this study suggest that ROS targeted therapy could be considered clinically in the treatment of HCC. They also mention that further studies of TBX15 within each cancer are needed to be focused on ROS concentrations and its signaling pathway to discover the true mechanism of tumor malignancy.
“Therefore, genome-wide DNA methylation profiling indicates that hypermethylation and reduced expression of TBX15 in tumor tissue represents a potential biomarker for predicting poor survival of patients with HCC.”
Click here to read the full scientific paper, published in Oncotarget.
—
Oncotarget is a unique platform designed to house scientific studies in a journal format that is available for anyone to read—without a paywall making access more difficult. This means information that has the potential to benefit our societies from the inside out can be shared with friends, neighbors, colleagues and other researchers, far and wide.
For media inquiries, please contact media@impactjournals.com.



